
Determination of the Apparent Dissociation Constants of Phosphoric Acid in SEAWATER1 | Semantic Scholar

Explain why there are different equilibrium constants, Ka1, Ka2, and Ka3 for phosphoric acid. | Homework.Study.com

The dissociation of weak electrolyte (weak acid) is expressed in terms of Ostwald dilution law. Stronger is the acid, weaker is its conjugate base. The dissociation constants of an acid (K(a)) and its conjugate base (K(b)) are related by the given relation : K(w ...

SOLVED: A solution of phosphoric acid, HyPO4, has molarity of 12.5 molar: Write the chemical equations for the step wise dissociation of phosphoric acid water: Calculate the concentrations of HyPO4, HzPOA HPO4

The values for pkas are given, can someone tell me which one corresponds to which form of the phosphoric acid? Thanks! : r/Mcat

Phosphorous acid Acetic acid Phosphoric acid Acid dissociation constant, salt, angle, text, logo png | PNGWing


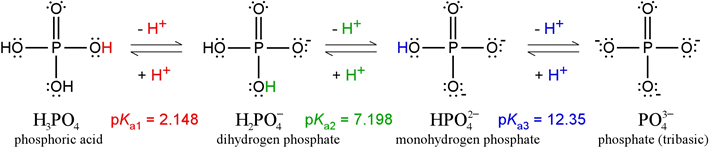



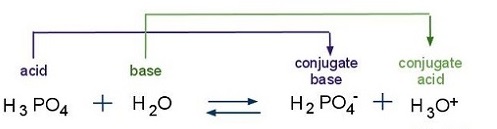



![Steps of H 3 PO 4 dissociation [8] | Download Scientific Diagram Steps of H 3 PO 4 dissociation [8] | Download Scientific Diagram](https://www.researchgate.net/profile/Ana-Bressiani/publication/250346512/figure/fig1/AS:669083511029765@1536533348163/Steps-of-H-3-PO-4-dissociation-8_Q320.jpg)




