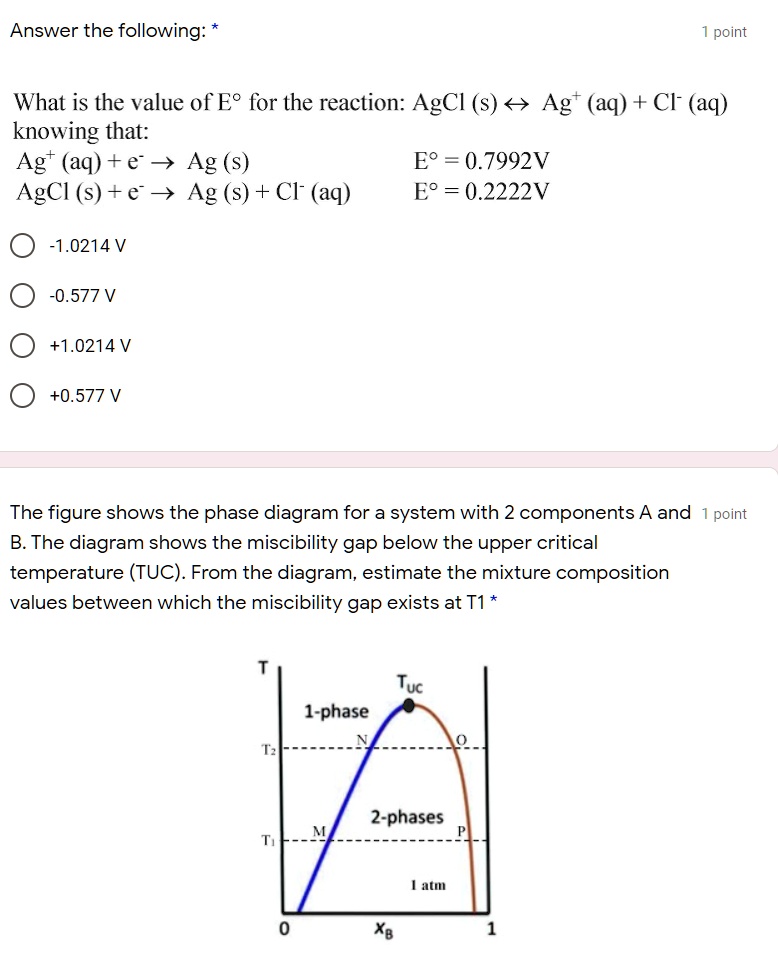
SOLVED: Answer the following: point What is the value of E? for the reaction: AgCl (s) < Agt (aq) + Cl (aq) knowing that: Agt (aq) + e v Ag (s) E? =

Miscibility Gap Closure, Interface Morphology, and Phase Microstructure of 3D LixFePO4 Nanoparticles from Surface Wetting and Coherency Strain | ACS Nano

Closed-Loop Miscibility Gap and Quantitative Tie-Lines in Ternary Membranes Containing Diphytanoyl PC: Biophysical Journal

Thermodynamic assessment of the miscibility gaps and the metastable liquidi in the Zn‐Bi, Zn‐Pb, and Zn‐Tl systems

Phase diagrams and critical temperatures for coherent and incoherent mixtures of InAs1−xSbx alloys using first-principles calculations: Journal of Applied Physics: Vol 131, No 21
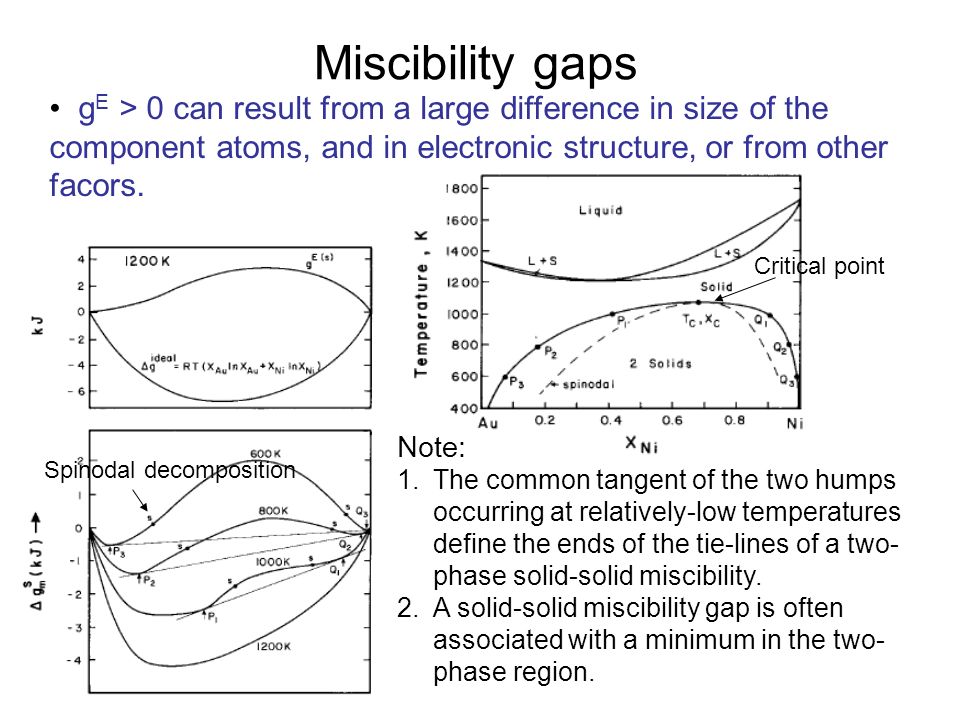
The shape of the two-phase lens typical of most metals Note: 1.Entropy of phase transformation is, in a decrease order, of vaporization, melting and solid-solid. - ppt download
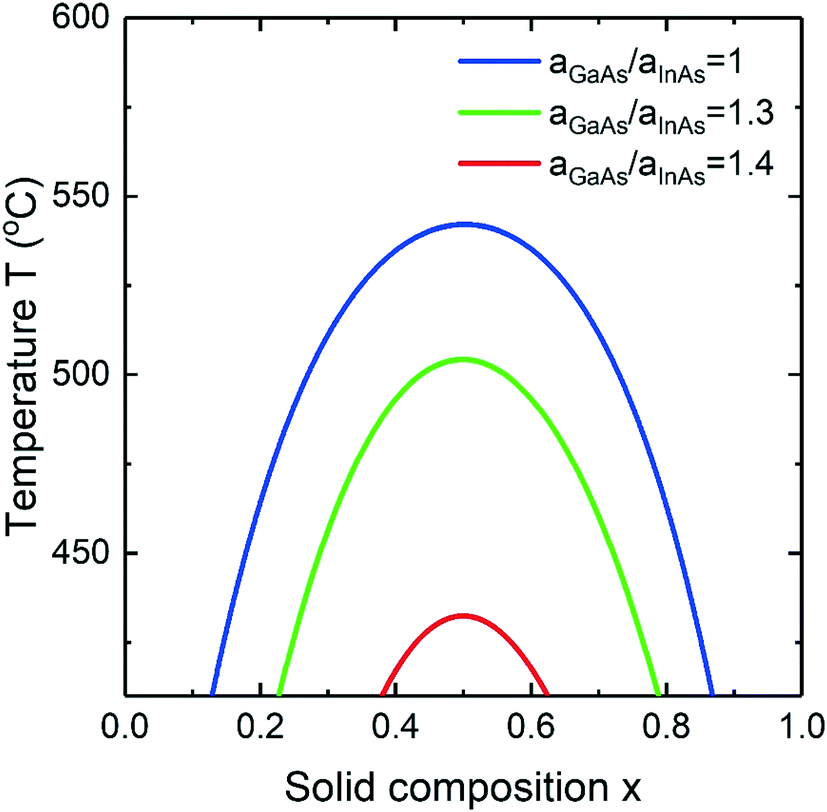
Surface energy driven miscibility gap suppression during nucleation of III–V ternary alloys - CrystEngComm (RSC Publishing) DOI:10.1039/D1CE00743B
The study of phase separation in the miscibility gap and ion specific effects on the aggregation of soft matter system