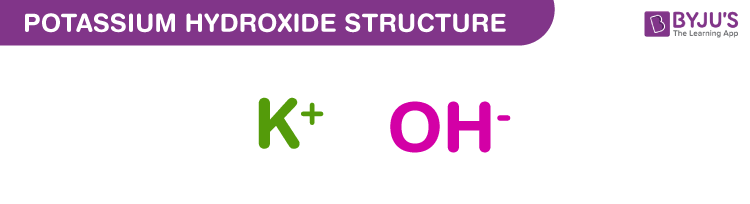
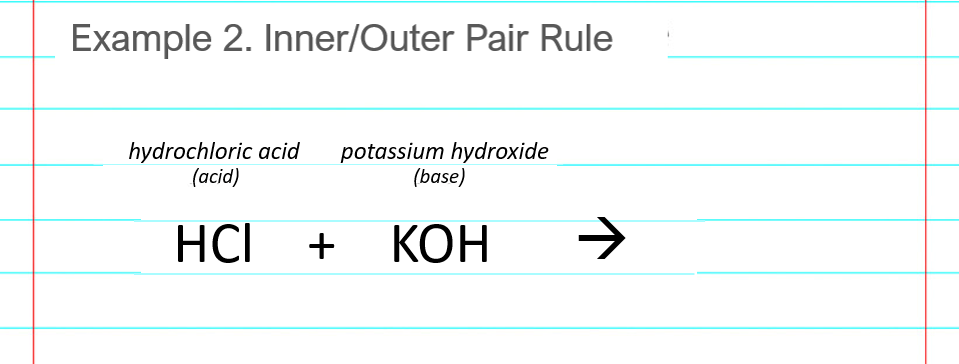
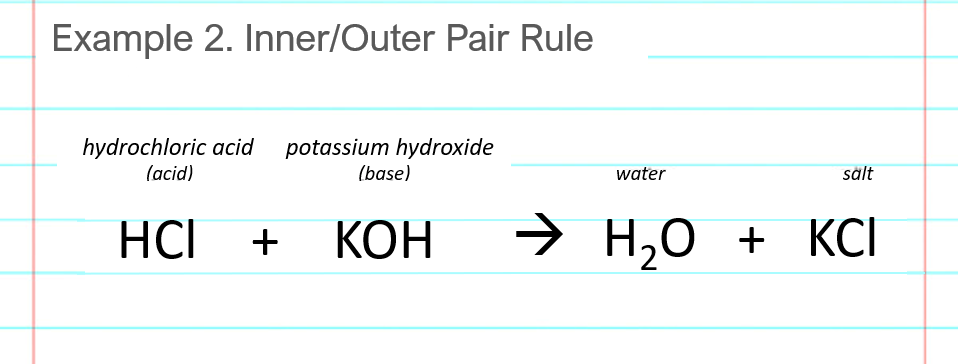
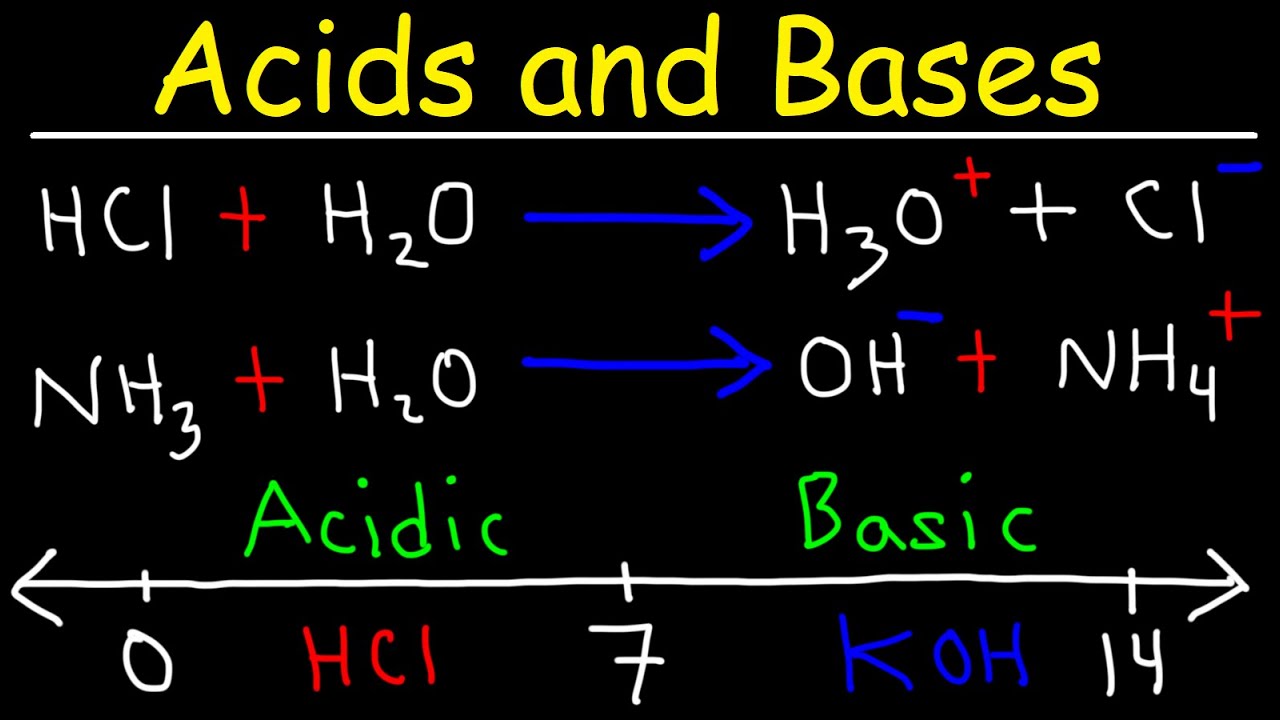
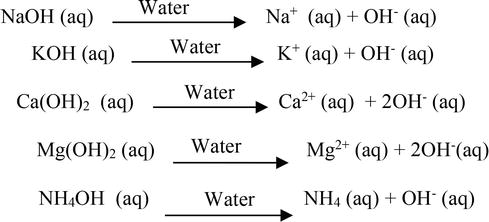
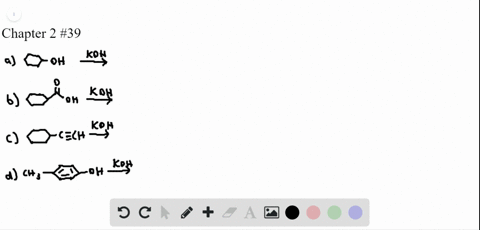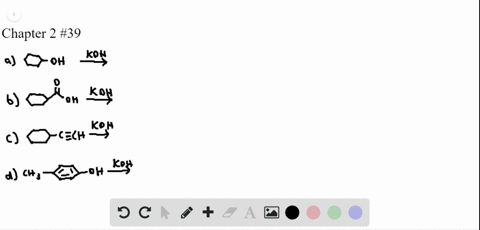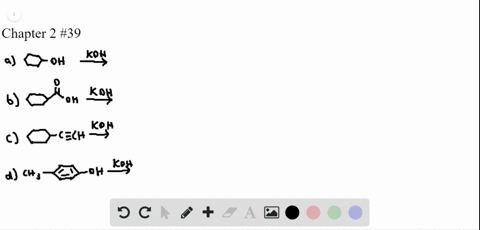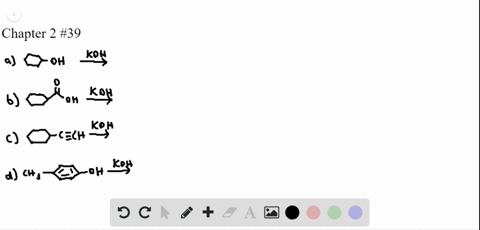
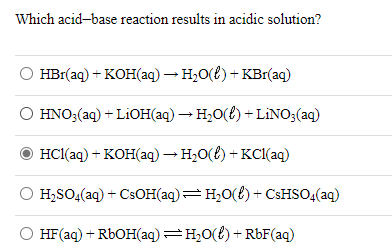Chapter 3: Acid – Base Equilibria HCl + KOH KCl + H 2 O acid + base salt + water. - ppt download

Write the neutralization reaction for the following acid and base: HCl_{(aq)} and KOH_{(aq)}. | Homework.Study.com

Write the full balanced chemical equation for the following acid-base neutralization reaction: HNO_3 + KOH to | Homework.Study.com

SOLVED: What is the products of this acid base reaction? HCI (aq) KOH (aq) KCI (aq) HzO () b) CIOH (aq) KH (s) CIOH (aq) KH (aq) KCI (s) HzO ()