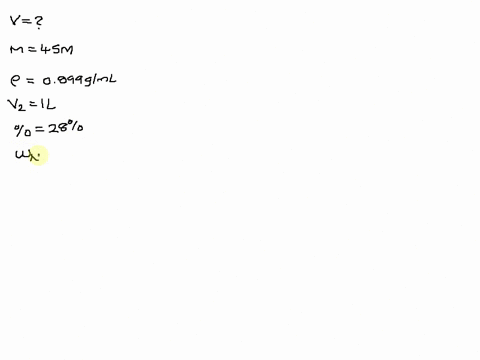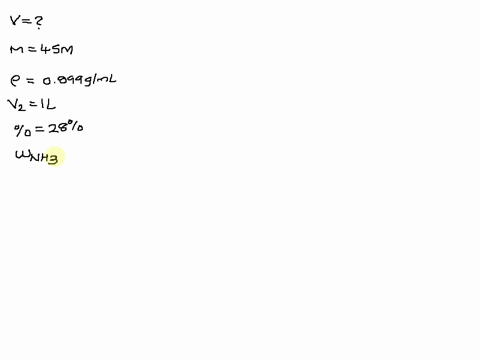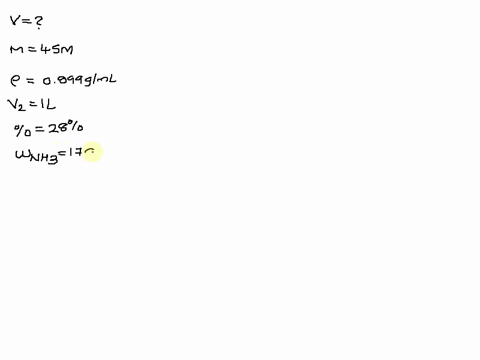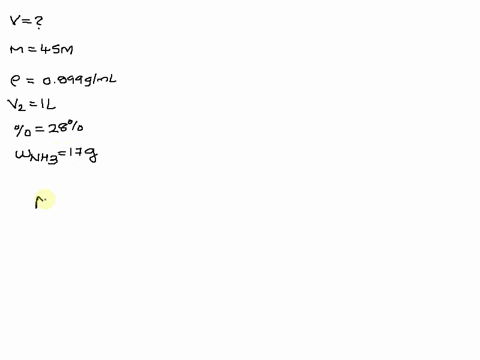SOLVED: The size of a certain insect population is given by P(t)equals100e Superscript . 03 t, where t is measured in days. At what time will the population equal 300?
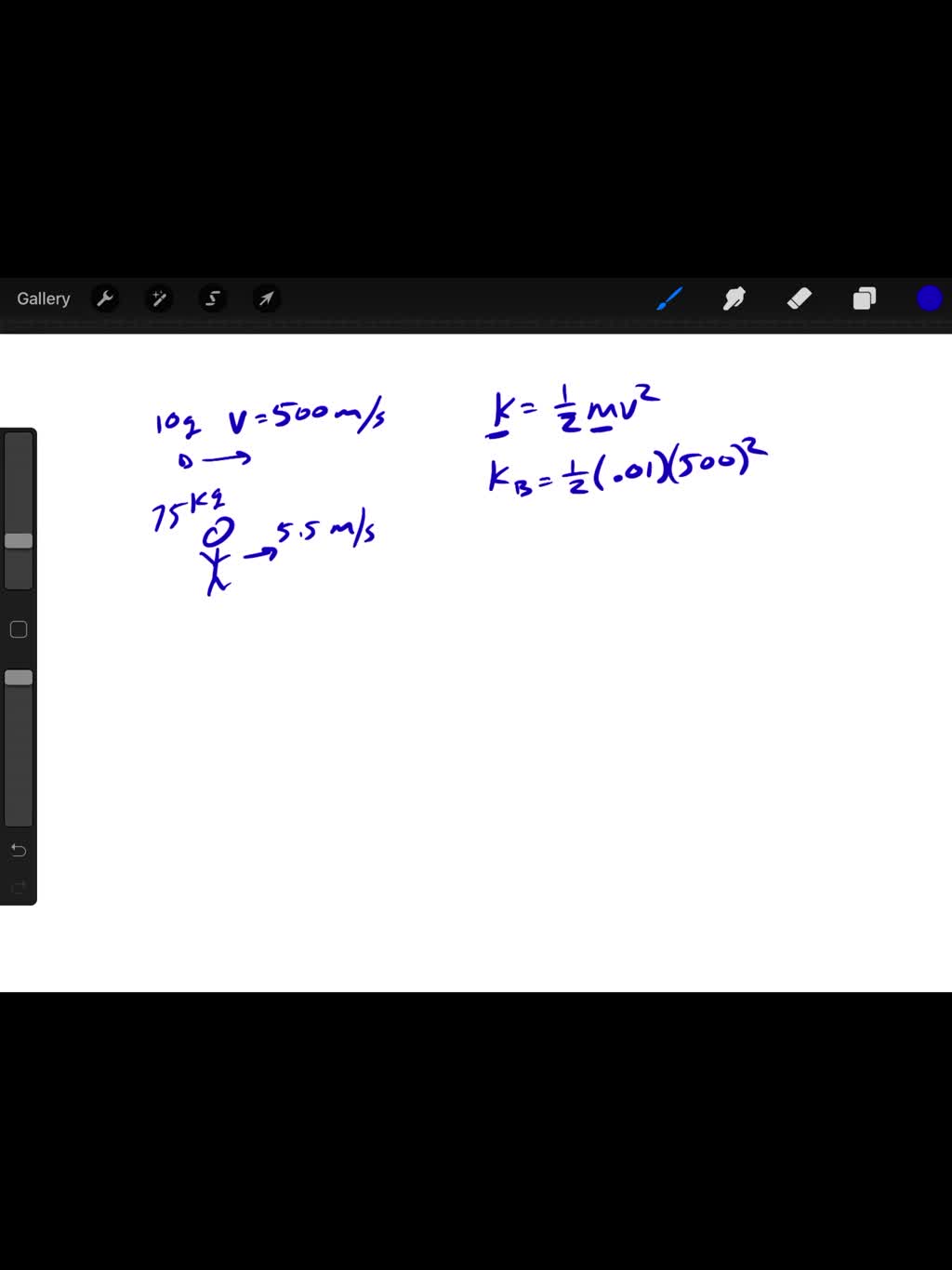
SOLVED:Which has the larger kinetic energy, a 10 g bullet fired at 500 m / s or a 75 kg student running at 5.5 m / s ?
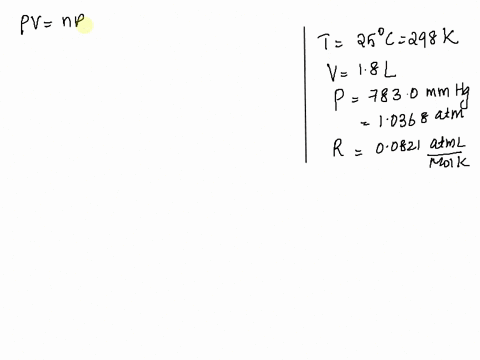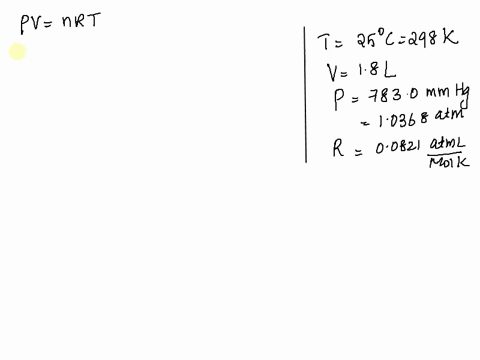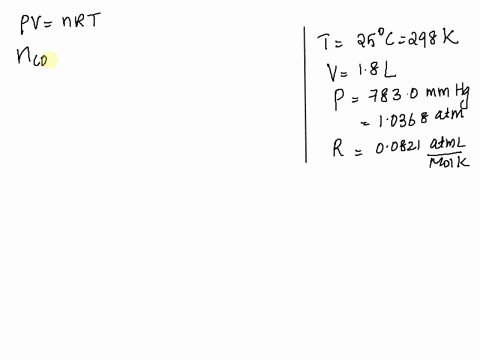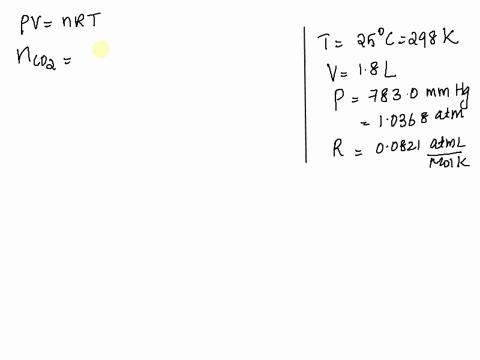
SOLVED: Given: Atmospheric Pressure, mmHg = 760. 0 mmHg Temperature of H2O, C= 19.5 € Vapor pressure of H2O at this temperature 17.0 mmHg Calculated pressure of CO2 in the system, mmHg=
![SOLVED: Determine [H3ot]in a solution where [OH] = 3.67 10 M Identify the solution as acidic; basic; Or neutral: [H3oh] = *10 L(Click to seleci) Solution is [(Click to selacu) SOLVED: Determine [H3ot]in a solution where [OH] = 3.67 10 M Identify the solution as acidic; basic; Or neutral: [H3oh] = *10 L(Click to seleci) Solution is [(Click to selacu)](https://cdn.numerade.com/ask_previews/47cd0432-84f1-482b-bdb9-3dbc55bb4925.gif)
SOLVED: Determine [H3ot]in a solution where [OH] = 3.67 10 M Identify the solution as acidic; basic; Or neutral: [H3oh] = *10 L(Click to seleci) Solution is [(Click to selacu)