
Elmira KHUSNUTDINOVA | PhD | Ufa Scientific Center of the Russian Academy of Science, Ufa | USC RAS | Research profile

IJMS | Free Full-Text | Novel A-Ring Chalcone Derivatives of Oleanolic and Ursolic Amides with Anti-Proliferative Effect Mediated through ROS-Triggered Apoptosis

Synthesis of A-ring quinolones, nine-membered oxolactams and spiroindoles by oxidative transformations of 2,3-indolotriterpenoids - ScienceDirect

Elmira KHUSNUTDINOVA | PhD | Ufa Scientific Center of the Russian Academy of Science, Ufa | USC RAS | Research profile

Synthesis and in vitro activity of oleanolic acid derivatives against Chlamydia trachomatis and Staphylococcus aureus | SpringerLink

Arthur Stanton was Army captain, well-regarded dentist | Herald Community Newspapers | www.liherald.com

Synthesis and alpha ‐ inhibitory activity of lupane type С2‐benzylidene‐ triterpenoids - Khusnutdinova - 2021 - Vietnam Journal of Chemistry - Wiley Online Library
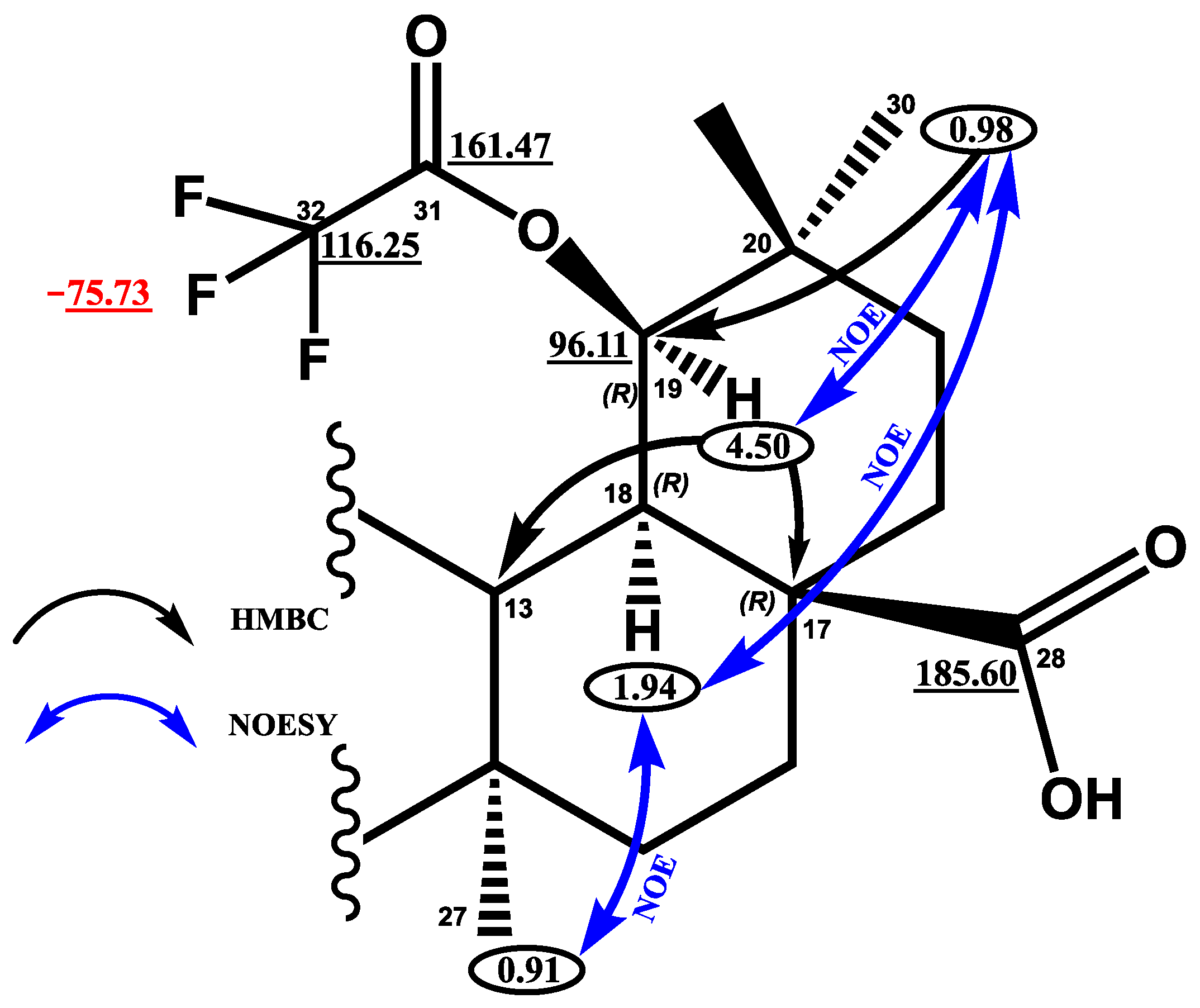
Molbank | Free Full-Text | Synthesis and Cytotoxic Potential of 3-Oxo-19β-trifluoroacetoxy-18αH-oleane-28-oic Acid

Elmira KHUSNUTDINOVA | PhD | Ufa Scientific Center of the Russian Academy of Science, Ufa | USC RAS | Research profile
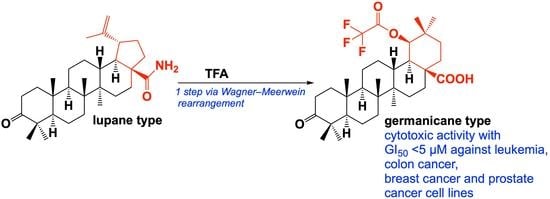
Molbank | Free Full-Text | Synthesis and Cytotoxic Potential of 3-Oxo-19β-trifluoroacetoxy-18αH-oleane-28-oic Acid
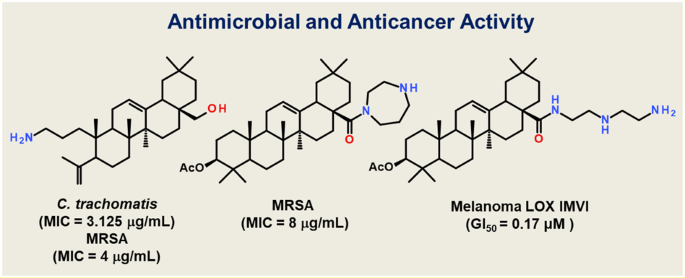
Synthesis and in vitro activity of oleanolic acid derivatives against Chlamydia trachomatis and Staphylococcus aureus | SpringerLink

Reagents and conditions: a PhNHNH2, AcOH, reflux, 15 h; b CH2N2, Et2O,... | Download Scientific Diagram

Elmira KHUSNUTDINOVA | PhD | Ufa Scientific Center of the Russian Academy of Science, Ufa | USC RAS | Research profile

Structure−Activity Relationship Anatomy by Network-like Similarity Graphs and Local Structure−Activity Relationship Indices | Journal of Medicinal Chemistry
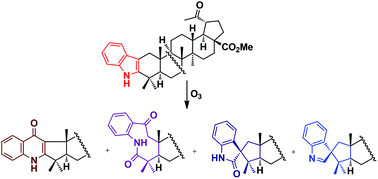
Synthesis of A-ring quinolones, nine-membered oxolactams and spiroindoles by oxidative transformations of 2,3-indolotriterpenoids - Organic & Biomolecular Chemistry (RSC Publishing)











