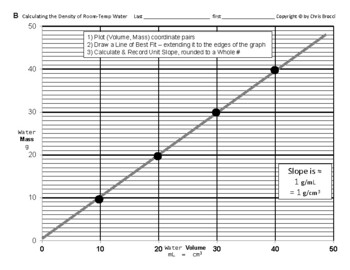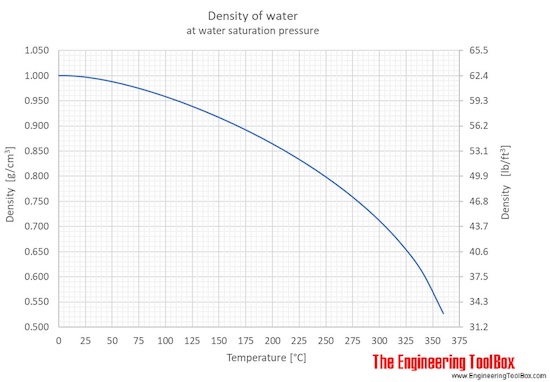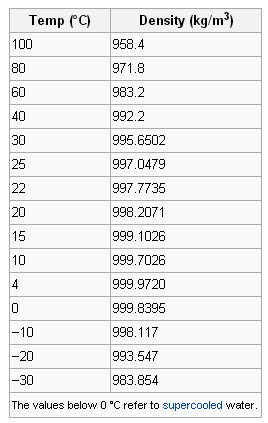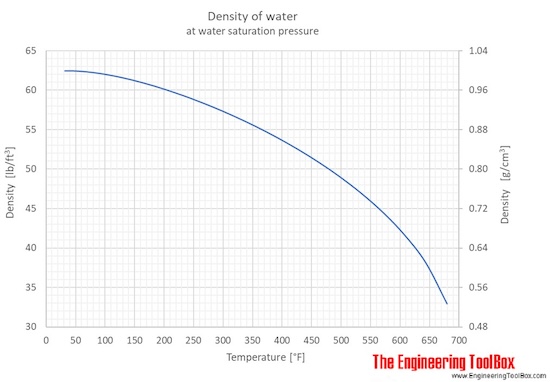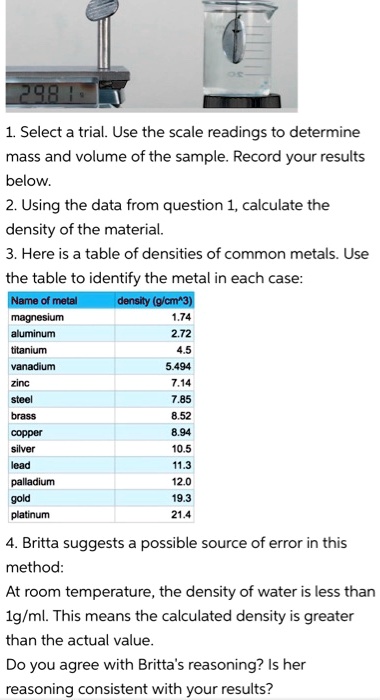
SOLVED: 98 1. Select a trial. Use the scale readings to determine mass and volume of the sample: Record your results below: 2. Using the data from question 1, calculate the density

the density of water at room temperature is 1.0 g/ml. how many molecules are there in a drop of water - Brainly.in

The density of water at room temperature is 1.0 g/mL. How many molecules are there in a drop of wate if its volume is 0.05 mL ?
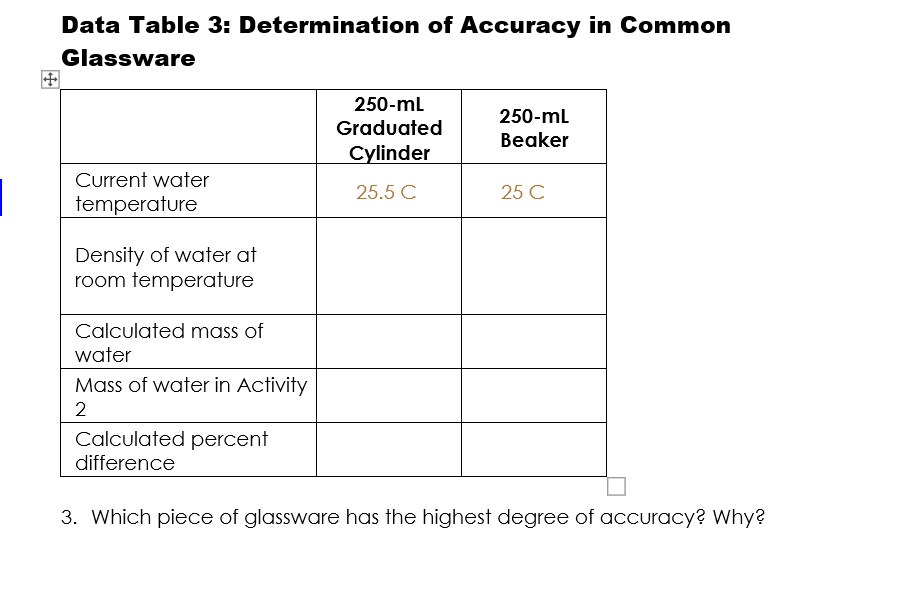
SOLVED: Data Table 3: Determination of Accuracy in Common Glassware 250-mL Graduated Cylinder 250-mL Beaker Current water temperature 25.5 € 25 € Density of water at room temperature Calculated mass of water

The density of water and ethanol at room temperature is 1.0 g / mL and 0.789 g / mL respectively. What volume of ethanol contains the same number of molecules as are
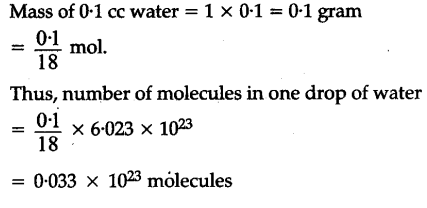
Density of water at room temperature is 1.0 g/ cc. How many molecules are there in one drop of water if its volume is 0.1 cc - CBSE Class 11 Chemistry - Learn CBSE Forum